The Cell Therapy CDMO Market is rapidly emerging as a transformative segment within the biopharmaceutical outsourcing industry, driven by the growing demand for advanced cell-based therapies. Contract Development and Manufacturing Organizations (CDMOs) play a critical role in supporting biotechnology and pharmaceutical companies by offering specialized services such as process development, manufacturing, testing, and regulatory support for cell therapies. These services enable companies to accelerate product development timelines while ensuring compliance with stringent regulatory standards.
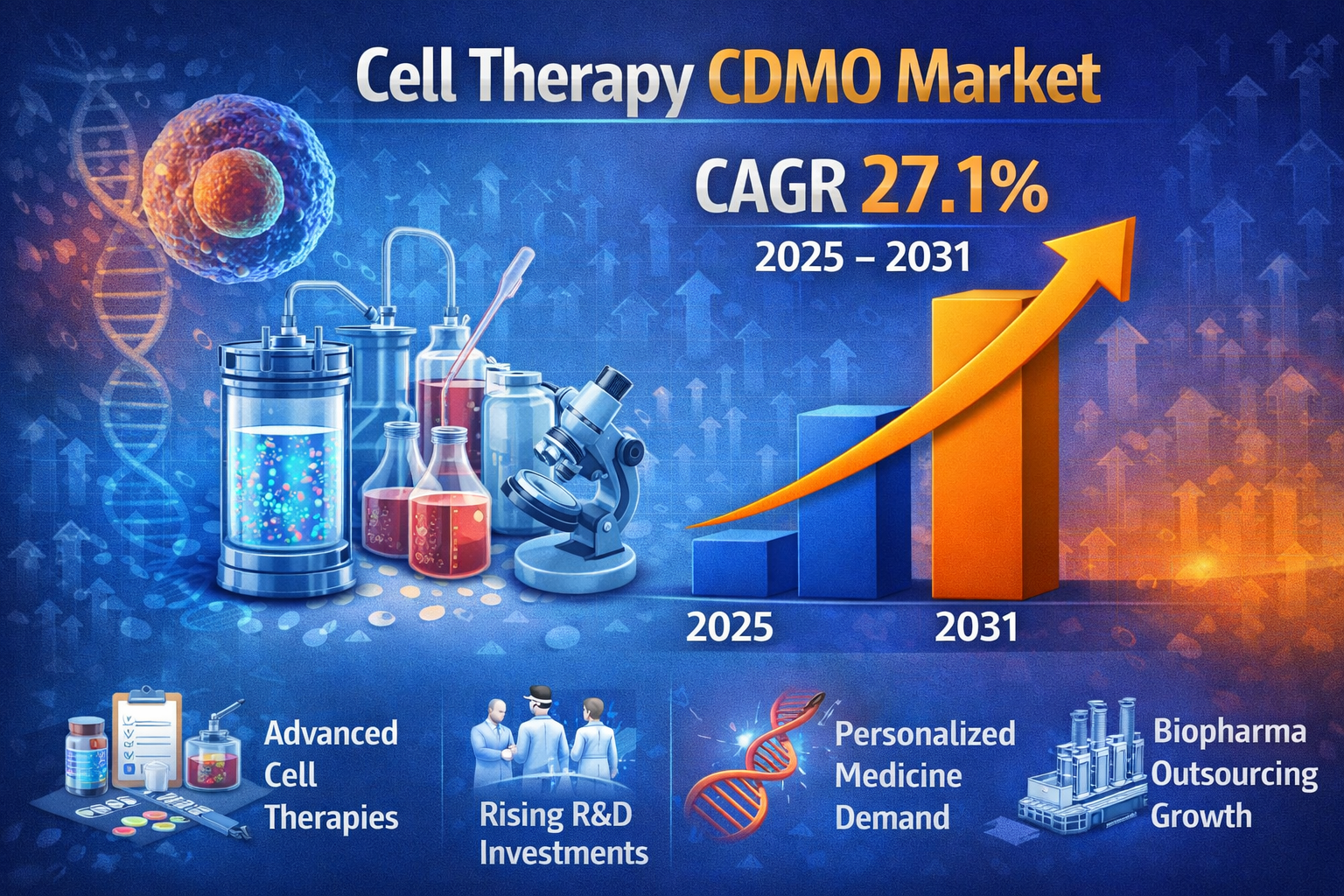
According to industry analysis, the Cell Therapy CDMO Market is projected to grow from US$ 4.12 billion in 2024 to US$ 21.92 billion by 2031, registering a remarkable CAGR of 27.1% during the forecast period from 2025 to 2031. This exponential growth reflects the increasing adoption of cell therapies, expansion of clinical pipelines, and rising outsourcing trends among biopharmaceutical companies.
Key Drivers of the Cell Therapy CDMO Market
The Cell Therapy CDMO Market is primarily driven by the rapid expansion of cell therapy pipelines and the increasing prevalence of chronic and rare diseases. Conditions such as cancer, autoimmune disorders, and genetic diseases are driving the demand for innovative therapies like CAR-T cell therapy and stem cell-based treatments. As pharmaceutical and biotech companies focus on developing these advanced therapies, the need for specialized CDMO services for manufacturing and development continues to rise significantly.
Another major driver of the Cell Therapy CDMO Market is the growing trend of outsourcing among biopharmaceutical companies. Developing in-house capabilities for cell therapy manufacturing requires substantial investment, advanced infrastructure, and highly skilled expertise. By partnering with CDMOs, companies can reduce operational costs, improve scalability, and accelerate time-to-market. Additionally, increasing regulatory complexity and the need for compliance with Good Manufacturing Practices (GMP) are encouraging companies to rely on experienced outsourcing partners, further fueling market growth.
Get Download Sample PDF : https://www.theinsightpartners.com/sample/TIPRE00040637
Technological Advancements Driving Market Innovation
Technological innovation is playing a pivotal role in shaping the Cell Therapy CDMO Market. Advanced manufacturing technologies, including automation, closed-system processing, and digital monitoring systems, are enhancing production efficiency and reducing contamination risks. These innovations are critical for maintaining the quality and consistency of cell-based therapies.
The integration of artificial intelligence (AI) and data analytics is also transforming the industry. AI-driven platforms enable better process optimization, predictive maintenance, and quality control, ultimately improving manufacturing outcomes. Additionally, advancements in gene editing technologies such as CRISPR and improved cell expansion techniques are enabling the development of more effective and scalable therapies.
Another key trend is the increasing adoption of allogeneic cell therapies, which offer scalability advantages compared to autologous therapies. CDMOs are investing in advanced infrastructure and capabilities to support large-scale production of these therapies, further driving market growth.
Emerging Opportunities in the Cell Therapy CDMO Market
The Cell Therapy CDMO Market presents substantial growth opportunities, particularly with the rising demand for personalized medicine. Cell therapies are inherently personalized, targeting specific patient needs and offering improved treatment outcomes. This trend is driving increased collaboration between biotech companies and CDMOs to develop customized therapeutic solutions.
Another significant opportunity lies in the expansion of clinical trials for cell-based therapies. The growing number of clinical studies focused on oncology and regenerative medicine is creating strong demand for CDMO services. As more therapies progress through clinical phases toward commercialization, the need for scalable manufacturing solutions is expected to increase.
Emerging markets in Asia-Pacific and Latin America also offer promising opportunities for growth. These regions are witnessing increased investments in biotechnology infrastructure, supportive government initiatives, and a growing number of research institutions. CDMOs expanding their presence in these regions can benefit from cost advantages and access to a large patient population.
Furthermore, strategic partnerships and collaborations between pharmaceutical companies and CDMOs are creating new avenues for innovation and market expansion. These collaborations enable knowledge sharing, resource optimization, and faster development of advanced therapies, positioning CDMOs as key enablers of the cell therapy ecosystem.
Regional Insights and Market Trends
North America currently dominates the Cell Therapy CDMO Market due to its strong biotechnology ecosystem, advanced research infrastructure, and high investment in cell therapy development. The presence of leading CDMOs and biopharmaceutical companies further strengthens the region’s market position.
Europe is another significant market, driven by increasing government support for advanced therapies and a growing number of clinical trials. Meanwhile, the Asia-Pacific region is expected to witness the fastest growth during the forecast period. Rapid advancements in biotechnology, increasing healthcare expenditure, and supportive regulatory frameworks are contributing to the expansion of the market in countries such as China, Japan, and India.
Leading Players in the Cell Therapy CDMO Market
Key companies operating in the Cell Therapy CDMO Market are focusing on expanding their capabilities, investing in advanced technologies, and forming strategic collaborations. Major players include:
• Lonza Group AG
• WuXi Biologics Inc.
• Thermo Fisher Scientific Inc.
• Catalent, Inc.
• Charles River Laboratories International Inc.
• AGC Biologics AS
• National Resilience Inc.
• Fujifilm Diosynth Biotechnologies
These companies are actively enhancing their manufacturing infrastructure and service portfolios to meet the growing demand for cell therapy outsourcing services.
Future Outlook
The future of the Cell Therapy CDMO Market appears highly promising, driven by continuous advancements in cell therapy research and increasing adoption of outsourcing models. The growing focus on personalized medicine, coupled with the rising prevalence of chronic diseases, will continue to drive demand for innovative cell-based therapies.
Additionally, technological advancements in manufacturing, increased investment in research and development, and expanding global collaborations are expected to accelerate market growth through 2031. Companies that prioritize innovation, scalability, and regulatory compliance will be well-positioned to capitalize on the significant opportunities in this rapidly evolving market.
Top Trending Reports:
Whole Cell Cholera Vaccine Market Overview, Growth, Trends, Analysis, Research Report (2026-2034)
Recombinant Hamster Ovary Cell (CHO) Hepatitis B Vaccine Market Report 2034 by Segments, Geography, Dynamics, Recent Developments, and Strategic Insights
Sickle Cell Disease Treatment Market Size and Competitive Analysis by 2030
About Us
The Insight Partners is a leading global market research and consulting firm that provides actionable insights across various industries, including healthcare, pharmaceuticals, biotechnology, and medical devices. Our reports are designed to help organizations understand market trends, identify growth opportunities, and make informed strategic decisions.
Contact Us
The Insight Partners
Email: sales@theinsightpartners.com
Phone: +1-646-491-9876
Website: www.theinsightpartners.com
Also Available in : Korean | German | Japanese | French | Chinese | Italian | Spanish